| Angiogenesis and Toxicology Research Lab |
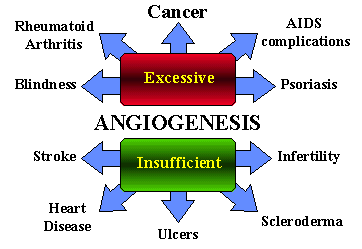
The term angiogenesis denotes the process of formation of new blood vessels from pre-existing small capillaries in the tissues and organs. This phenomenon accompanies some physiological processes such as wound healing, morphological changes observed within the female reproductive organs during the menstrual cycle, and formation of the placenta in pregnancy. Excessive formation of blood vessels can also be observed in a whole range of pathological processes connected with the development of rheumatoid arthritis, diabetic retinopathy, endometriosis, psoriasis and juvenile angiomas. In 1963 Folkman observed that neovascularization occurs during the formation and growth of solid tumors. The formation of new blood vessels within neoplasms, which provides the tumor tissue with oxygen and basic energetic compounds, is a complex process. It involves not only mutated tumor cells but also endothelial cells, the basal membranes of the nearest capillaries and the stroma of the neoplasm. This fundamental observation has become a stimulus for further studies on the role of angiogenesis in the course of carcinogenesis. Cancer tissue is highly vascularized. In recent decades, this observation has led to the realization that blood vessels are essential for tumor growth, invasion, and metastasis. Indeed, developing solid primary tumors are thought to remain clinically insignificant, with maximum sizes no greater than about 2 mm, unless they can arrange to steal nourishment from their host.
Biochemically, an invasive tumor acts by altering what now appears to be an elaborate balance of factors affecting vascular endothelial cells, thereby inducing new vascular networks to spring from existing venules or capillaries.
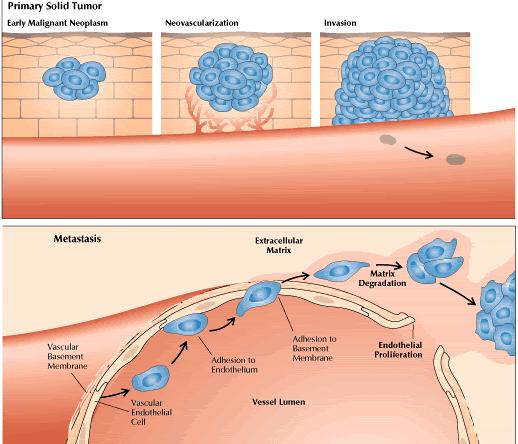
By the late 1980s, the first agents targeting tumor-related angiogenesis were entering phase I clinical trials. Today, drugs with action at several biomolecular targets are being studied. Some of them inhibit growth factors that promote endothelial proliferation, whereas others inhibit the proteases required for endothelial cells to penetrate basement membrane and form new blood vessels. Some of the newest agents alter specific intracellular signal transduction pathways. Others act in ways that remain obscure. So far, clinical trials have found the agents to be well tolerated, with minimal side effects. Overall, the drugs are predicted to be chiefly cytostatic rather than cytotoxic; they stabilize tumors and perhaps prevent metastasis rather than cure. On the other hand, it remains possible that these agents may trigger apoptosis (programmed cell death) and thereby promote tumor regression. Just as the target is novel, the drugs are likely to exhibit novel antitumor effects. Hence, novel evaluative strategies will be required to define the treatments' potential roles in clinical oncology. To predict what angiogenesis inhibition might achieve against cancer, one must survey the ways in which angiogenesis serves a tumor at various stages of its development (Figure 1). Initially, through mutation, a cell escapes the normal constraints on its growth. In particular, a protooncogene may become active or a tumor suppressor gene inactive, permitting proliferation. While the resulting clone is still minute, angiogenesis is not essential. The tumor cells receive oxygen and nutrients by diffusion.
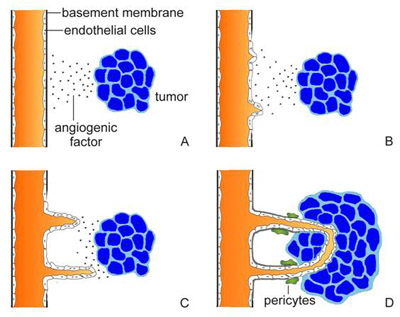
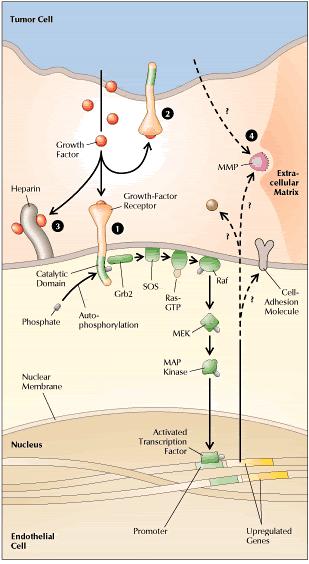
Once the tumor reaches a diameter of about 2 mm, however, it requires its own blood vessels for attainment of greater size. Thus, further growth triggers capillary formation, a process normally suppressed. Evidence increasingly suggests that control of a host angiogenic switch is often acquired early in tumorigenesis. In fact, in several animal models of human tumor xenografts, neovasculature has been detected at stages that may precede the first appearance of a discrete neoplasm. The induction of neoangiogenesis is a complex and varied process. In some instances, a genetic event promoting tumorigenesis may itself promote vascularization. In cultured fibroblasts, for example, loss of the tumor suppressor gene p53 causes reduced production of thrombospondin-1, an endogenous angiogenesis blocker. The implication is that suppression of neovascularization is a facet of the tumor suppressor gene's capacity to inhibit a malignant phenotype. In other cases, proangiogenic mutations may arise from the genetic instability of cancer cells. Thus, in a cascade of genetic events, a cell may undergo a mutation that upregulates expression of a growth factor for endothelial cells, instituting a paracrine loop involving VEGF (vascular endothelial growth factor) or bFGF (basic fibroblast growth factor). Many such growth factors are multifunctional (Figure 2). Hence, the same factor that stimulates blood-vessel development may also upregulate expression of the proteolytic enzymes required for tumor expansion. Endothelial cells responsive to the growth factor may lie within the tumor mass. Alternatively, paracrine signals may influence adjacent endothelium.
Sometimes the tumor cells themselves become highly responsive to an angiogenic growth factor. For example, a tumor cell may exhibit upregulation of both VEGF and a VEGF receptor, thereby creating an autocrine signal loop. The degree of growth-factor responsiveness varies sharply from one tumor type to another, and even among individual tumors. Kaposi's sarcoma is often highly growth-factor-dependent and also highly angiogenic. For other tumor types, such characterizations have been elusive. However, angiogenic growth factors, including VEGF and bFGF, seem to be commonly expressed among many tumor types. In brief, neovascularization appears to involve a dual effect in which angiogenic growth factors may be produced by and may stimulate both tumor cells and endothelial cells. Likewise, adhesion molecules expressed on both types of cells may promote blood vessel formation. Histopathologically, neoangiogenesis begins with local degradation of extracellular matrix or basement membrane, enabling endothelial cells to invade the stroma and migrate. At the molecular level, the process is believed to require not only proteases and growth factors but also cell-surface adhesion molecules and various constituents of the extracellular matrix. At the head of the migrating column, cells proliferate; behind, cells differentiate and organize into tubes, which finally coalesce into loops. The same characteristics required for tumor vascularization are essential for tumor spread. At the primary tumor, degradation of extracellular matrix facilitates local tissue invasion. Additionally, the degradation combines with the leakiness of tumor neovasculature to enhance the opportunities for tumor-cell entry into blood vessels. At distant host tissues, degradation of extracellular matrix assists extravasation. Lodged in the alien environment of a distant organ's parenchyma, cancer cell demonstrate their malignant potential by utilizing proteases, growth factors, and extracellular contacts to invade tissue and stimulate further angiogenesis. The main aim of our lab is to screen the effects of different environmental toxicants on angiogenesis and also to study the effects of different indigenous plants to find suitable candidates for cancer treatment. |